The molecular geometry of PF3 is a trigonal pyramid and its electron geometry is tetrahedral because phosphorous has Sp³ hybridization with 5 valence electrons in its valence shell and it makes three bond pairs one with each fluorine atom. We start by determining the hybridization of each carbon atom.

Pf3 Lewis Structure Molecular Geometry Polar Or Nonpolar Bond Angle

Pf3 Molecular Geometry Shape And Bond Angles Youtube

Why Is The Molecular Geometry Of Pf3 Trigonal Pyramidal Phosphorus Trifluoride Quizalize
BrF3 or bromine trifluoride is a highly polar autoionizing solvent.

Pf3 molecular geometry. Molecular Geometry of Sulfur Trioxide SO3 The above image clears that the bond angle among oxygen-sulfur-oxygen O-S-O atoms have to be more than 90. Is the molecule polar. Molecular Geometry and Bonding Theories.
BrF3 Lewis Structure Bromine Trifluoride. 4 The Chemistry of Sulfur N. The molecular geometry of ClF3 is T-shaped and electron geometry is Trigonal bipyramidal.
H2co resonance structures. Like PF3 and PCl3 PBr3 also exhibits the properties of both Lewis Acid and Lewis Base. As well as the molecular geometry and bond anglesFor the PH4 structure.
Polarity does not apply to polyatomic ions 1 PF3. The molecular geometry is the shape of the molecule. B 1 lone pair bent E 3 lone pairs linear.
It aims to help students hone their analytical and problem-solving skills by presenting detailed approaches to solving chemical problems. Determine the electron geometry molecular geometry and polarity of HBrO2. Lewis structure of.
Cs3n ionic or covalent. Northern Arizona University and Raymond Chang this success guide is written for use with General Chemistry. 30 Determine the electron geometry molecular geometry and polarity of SF6.
A 0 lone pairs linear D 3 lone pairs bent. A egtrigonal bipyramidal mgtrigonal bipyramidal nonpolar B egtetrahedral mgtetrahedral polar C egtrigonal bipyramidal mgsee-saw polar D egoctahedral mgtrigonal bipyramidal nonpolar E egoctahedral mgoctahedral nonpolar. Take A Sneak Peak At The Movies Coming Out This Week 812 Best Reactions to Movies Out Now In Theaters.
BrF3 Molecular Geometry Bond AnglesBromine Trifluoride Bromine Trifluoride or BrF3 is an. Because of 5 regions of. SBr2 molecule has a tetrahedral or V-shaped bent molecular geometry because there is an electrical repulsion between the lone pairs of electrons in sulfur and two single bond pairsS-Br of the.
Egtetrahedral mgbent polar Choose the compound below that contains at least one polar covalent bond but is nonpolar. SO3 has an fill in the blank electron domain geometry and an fill in the blank molecular geometry. For each molecule on the worksheet the.
PF3 Phosphorus Trifluoride PH3 Phosphine PI3 Phosphorus Triiodide PtNH32Cl2 Diamminedichloroplatinum RaOH2 Radium Hydroxide RbOH Rubidium Hydroxide S2F10 Sulfur Fluoride SbCl3 Antimony Tricloride SCl6 Sulfur Hexachloride SF4 Sulfur Tetrafluoride SF6 Sulfur Hexafluoride SiH4 Silane SiO2 Silicon Dioxide SiO2 Quartz SnCl4 TinIV Chloride SnF4. What is the molecular geometry of PF3. Lewis electron dot structure Molecular geometry b Predict with a reason whether the molecule PF3 is polar or non-polar.
SBr2 has dot electron representative structure. From the overall molecular geometry and the presence and arrangement of polar bonds if any determine if a molecule is polar. Give the number of lone pairs around the central atom and the molecular geometry of XeF2.
Structure the number of valence electrons the electron arrangement ea and the molecular geometry mg are given click on the rotating molecule for summary of. Aug 21 2021 CH2O has a molecular geometry of AX3 trigonal planar shape and an sp2 hybridization. It binds to the iron present in the blood hemoglobin to spread throughout the body and prevents the blood from absorbing the oxygen.
You may use the tables at the back of the book. From aluminum to xenon we explain the properties and composition of the substances that make up all matter. The molecular geometry of NO2- is Use VSEPR to justify your answerA bent bond angle - 109B trigonal planarC linearD bent bond angle - 120 Predict the ideal bond angles around each central atom in this molecule.
014 EC Number 234-129-0 PubChem CID 25353 RTECS number WS4500000 UNII 8Q6684WQ9H Y UN Jan 16 2015 SCl_2 has a bent molecular geometry with bond angles of approximately 103 and a bond. The total valence electron present available for drawing the ClF3 Lewis structure is 28 According to the ClF3 Lewis structure 2 lone pairs and 3 bonded pairs are present in this molecule. Furthermore the angles are not exactly 120 degrees as in the case of a trigonal planar molecule where the surrounding atoms are all the same.
C 2 lone pairs bent Ans. Give the number of lone pairs around the central atom and the molecular geometry of SCl2. Aluminum p 4 13 n0 5 14 e 4 13 valence e 2 3 core e 2 10.
Is C2H5OH Polar or Nonpolar. Academiaedu is a platform for academics to share research papers. PF3 Lewis Structure Molecular Geometry and Hybridization Phosphorus trifluoride PF3 is colorless as well as an odorless gas having similar toxicity as that of carbon monoxide.
Moreover through the valence shell electron pair repulsion VSEPR theory the structure of sulfur trioxide SO3 is found to be bent shaped or trigonal pyramidal or trigonal planar where the bond angle is 120. New Movie Releases This Weekend. 1 Chemical Bonds Lewis Symbols and the Octet Rule.
The molecular geometry of BeBr2 is linear with symmetric charge distribution on the central atom. Valence Electrons And Lewis Dot Structure Worksheet Answers Printable Worksheets www. One may also ask is BrF3 polar or non polar.
Molecular geometry is the three-dimensional arrangement of atoms in the molecules that aids us to understand the structure of the molecule better. To be empirical a formula must be incapable of being reduced.
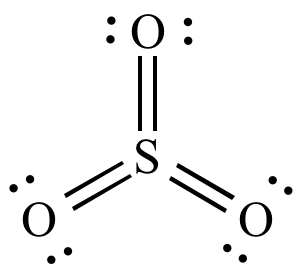
Compare The Structures Of So3 To Pf3 And Explain Why They Have Different Molecular Shapes Socratic
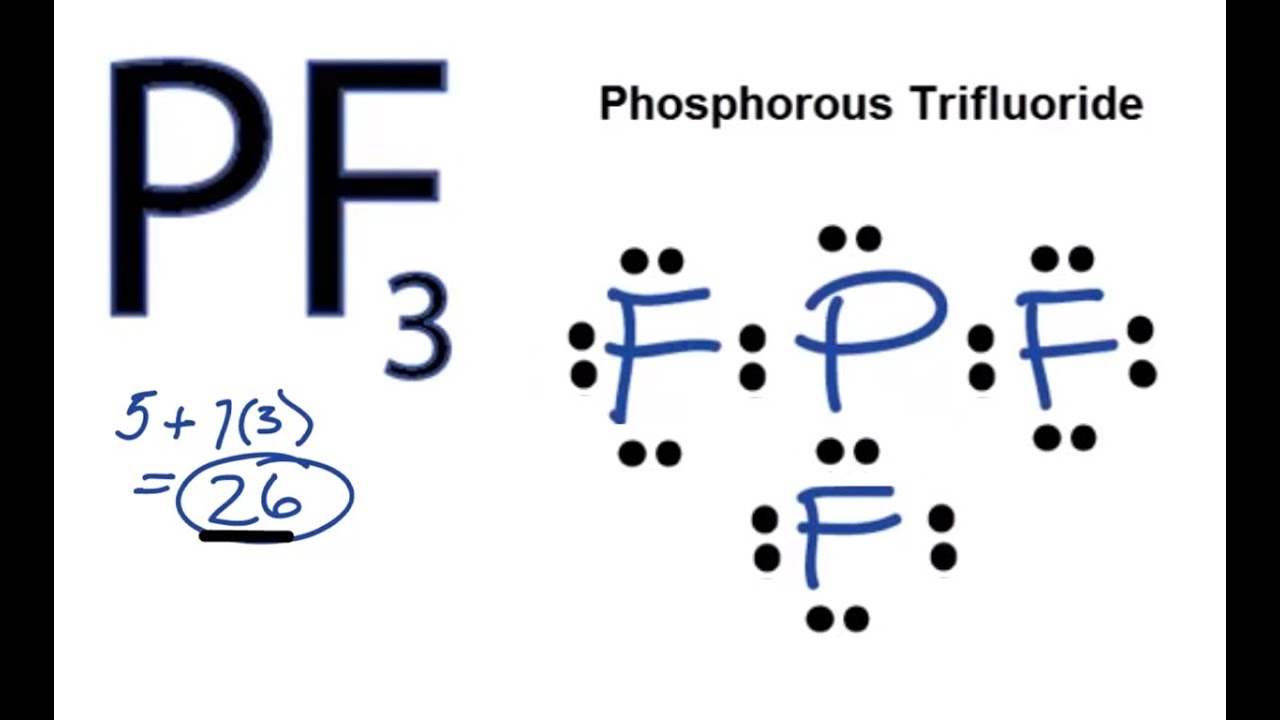
Pf3 Lewis Structure Molecular Geometry And Hybridization Techiescientist

Dublin Schools Lesson Molecular Geometry What Shapes Do Molecules Have
Short Question T4 Chemhoper

Get Answer Pf3 Draw The Lewis Structure For Pf3 In The Box At The Right Transtutors
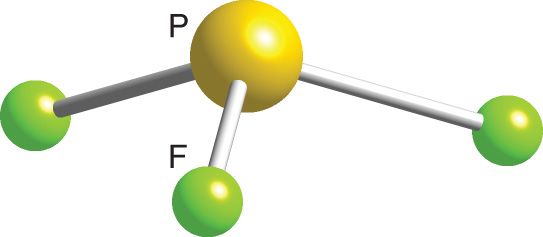
Phosphorus Trifluoride Pf3

Phosphorus Pentafluoride Wikipedia

Pf3 Molecular Geometry How To Discuss
